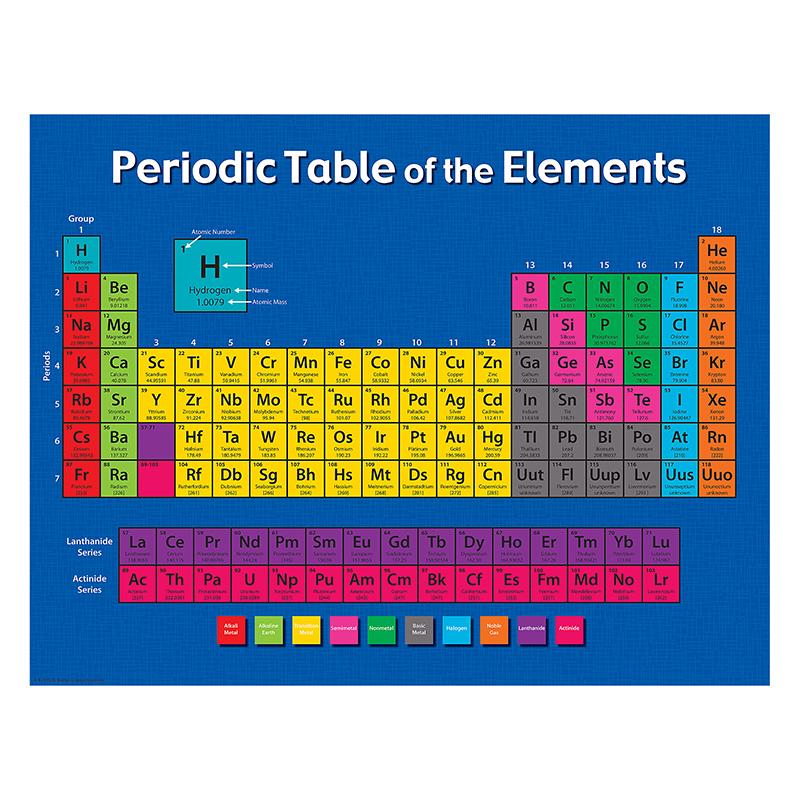
Mendeleev wasn’t the first to come up with a system to categorize elements, but, Scerri points out, “his version is the one that had the biggest impact on the scientific community.” The periodic table organizes elements by rows according to their atomic number, the number of protons in an atom’s nucleus, and by columns according to the configurations of the atom’s outermost electrons. In the beginning, not even Mendeleev recognized the magnitude of his discovery. It captures the essence of chemistry in one elegant pattern.” Dmitri Mendeleev first published a scheme to organize all then-known elements in 1869, and this system, although not perfect, became fundamental in the study of chemistry. “It is one of the most powerful icons of science. “It graces the walls of lecture halls and laboratories of all types, from universities to industry,” he wrote in American Scientist. The periodic table of elements is a “stalwart symbol” of chemistry, the chemist Eric R. Efforts to find the next elements, 119 and 120, are underway.

The new elements also received their final names: nihonium, moscovium, tennessine, and oganesson.
But as we enter the International Year of the Periodic Table, the classic periodic table has been filled to its seventh row: In late 2015, the International Union of Pure and Applied Chemistry officially confirmed elements 113, 115, 117, and 118. Until December 2015, there were holes in the periodic table, elements synthesized but not yet officially recognized.
